Recall that waves are characterized by several factors: speed, wavelength, frequency, energy, etc. The speed of a wave tends to be characterized by the material, or medium, through which the wave is propagating. (Remember, for example, that the speed of a wave on a string depends on the tension along with the mass-per-unit-length of the string.) Electromagnetic waves are strange (and unique!) in that they do not need any medium through which to propagate they can travel through otherwise empty space (although they can also travel through various media). Nevertheless, the speed of EM radiation depends on the medium through which the light is propagating. (This important phenomenon will explain why light can be focused by lenses, for example.)
Given the speed of the EM radiation within a given medium, the wavelength and frequency are related by the famous equation (which is true for all types of waves, not just EM waves!) speed = wavelength times frequency, or
If the light is traveling in a vacuum (empty space), then the speed of the light is c = 3.0 x 10 8 m/s, and we would write
As the light changes from one medium to another, the frequency of the wave remains a constant only the wavelength and speed change. In the following discussion, all wavelengths are given for EM radiation traveling through empty space.
We will find out later in this course that the energy of an EM wave is determined by its frequency (or wavelength) the greater the frequency, the greater the energy carried by the wave.
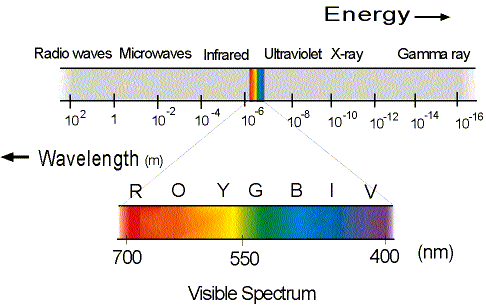
The wavelengths (or equivalently, frequencies) of EM radiation are broken up into various regions, none of which are precisely defined. The two broadest categories are visible and nonvisble light. (By visible, we mean, of course, visible by the human eye.) The various wavelengths (or frequencies) of EM waves form what is called the electromagnetic spectrum. The visible region of the electromagnetic spectrum normally extends from about 390 nm to about 780 nm. (Recall that a nanometer, nm is equal to 1 nm = 1 x 10 9 m.) For simplicity, we will take the visible region of the EM spectrum to be from about 400 nm = 4.0 x 10 7 m (violet light) up to about 700 nm = 7.0 x 10 7 m (red light). Again, these wavelengths are not precisely defined in that light that is 640 nm may also look red to you. Where the light stops looking red and starts look orange can vary from person to person. (Some people have reportedly been able to see light with wavelengths as small as 310 nm or as large as 1050 nm! See the table below for accepted values of the wavelength ranges for the various colors in the visible spectrum.)
Wavelengths just shorter than 400 nm are said to be in the ultraviolet region of the spectrum (yes this is the region responsible for sunburn...). This region extends from about 10 7 to 10 8 m. As we get to even shorter wavelengths, we start getting into the x-ray region of the EM spectrum, which is around the 10 9 10 11 m. The gamma ray region of the EM spectrum extends from about 10 13 to 10 16 m, a region which corresponds to very high frequencies and thus very high energies.
At the other side of the visible region of the EM spectrum that corresponding to wavelengths larger than those of visible light we have first of all the infrared region just beyond the red end of the visible spectrum. The infrared region extends from about 10 6 to 10 3 m. Microwaves have wavelengths from about 10 3 up to about 10 1 m. (This region of the EM spectrum accounts for most of the EM radiation found in the universe.) Finally, the radio wave region of the EM spectrum extends very roughly from about 10 1 m up to 10 2 m = 100 m.
Again, the wavelengths given above correspond to EM radiation in a vacuum and are rough guidelines the cut-offs for the various regions of the spectrum are not well defined.
As mentioned above, the visible regionof the EM spectrum extends from about 400 nm to about 700 nm. (Even this range varies from person to person!) The different colors of the visible region of the spectrum correspond to different wavelengths (or frequencies) of radiation. The following wavelengths are at about the middle of the band of wavelengths corresponding to the given colors (all are approximate!): violet 420 nm; indigo 440 nm; blue 470 nm; green 530 nm; yellow 580 nm; orange 610 nm; red 660 nm.
You are not expected to memorize the exact numbers given above (since the numbers given above are not exact anyway!). But you should be familiar with the approximate regions of the spectrum and the various colors of the visible region of the spectrum. Many generations of physics students have remembered the approximate wavelengths of the colors of the visible spectrum by simply remembering the (odd!) name Roy G. Biv. The letters, of course, stand for Red-orange-yellow Green. Blue-indigo-violet, the colors listed in reverse order from the list given above. If you write out wavelengths from 700 nm to 400 nm in steps of 50 nm (that is, write out the list 700 nm 650 nm 600 nm 550 nm etc.), and then arrange the letters ROYGBIV above the numbers spaced out evenly above them with yellow and green to either side of 550 nm (the middle of the region), then you will have the approximate wavelengths of the various colors, as shown in the lower portion of Fig. 1.1 below.
The various regions of the electromagnetic spectrum and the wavelengths of the colors in the visible region of the spectrum are shown in Fig. 1.1 below.
|
It should be noted that the method given above for approximating the wavelengths of various colors in the spectrum (Red, Orange, Y ellow, etc.) is very approximate and not quite accurate in some wavelength regions. Nevertheless, we will use this method when we are interested in only a rough idea of the wavelength of a given color, or the color corresponding to a given wavelength.
It is interesting to note that there is no universally agreed-upon wavelength or range of wavelengths corresponding to each of the colors in the spectrum. It is of course also true that the perception of color of a specific wavelength can vary from person to person. In addition, as if that werent bad enough, the perception of color can vary with intensitya wavelength that appears to be red at a low intensity (or brightness) may appear to be orange at a higher intensity.
The table below gives an approximate range of wavelengths corresponding to the various colors in the visible spectrum. While these are not universally agreed-upon values, they nevertheless tend to more-or-less agree with the various wavelengths found in the literature. The values in this table are given only for your referenceyou should not feel obligated to memorize them!
|
||||||||||||||||||||||||||||||||||||
Light is detected in the human eye by the rods and cones on the retina (the back lining in the eyethis is discussed in Lecture 3 on the Optics of the Human Eye). The colors of light are perceived by means of the stimulation of three different types of cones: one most sensitive to blue, one most sensitive to yellow-green, and one most sensitive to red-orange. The apparent color of an object is a result of the degree of stimulation of these three types of cones. The eye is most sensitive to yellow light. It is due to these factors that the sun, for example, appears to be yellow, despite the fact that it gives off wavelengths throughout the entire visible (and invisible!) spectrum, and gives off the most radiant energy within the green to yellow-green portion of the visible spectrum. (See Lecture 6 on Blackbody Radiation.)
We note in passing that another common unit of wavelength is the Angstrom: 1 Angstrom = 1 Å = 10 10 m. Thus, for example, 400 nm = 4,000 Å = 4.0 x 10 7 m. The symbol for an Angstrom is Å (a capital A with a small circle above it, a letter in the Swedish alphabet). We will tend to use nanometers for visible-light wavelengths in this course.