We have seen that a solution of the Schrödinger equation for the hydrogen atom results in wavefunctions that describe a probability of finding the electron at various positions and times around the nucleus. We saw in the last section that the size of the atom tends to increase as the principal quantum number n increases (rn = n2 r1, Eq. 9.3).
The various quantum states not only have different sets of quantum numbers and different wavefunctions (or probability functions), but they also have different energies. It turns out (from the solution of the Schrödinger equation) that the energy of an electron in an isolated hydrogen atom (one that is not interacting with other atoms or electric or magnetic fields) depends only on the principal quantum number n of the quantum state occupied by the electron. In particular, the energy of the electron in a hydrogen atom is given by
where m is the mass of the electron (m = 9.11 x 10 31 kg), e is the electron-charge magnitude (e = 1.6 x 10 19 C), eo is a constant called the electric permittivity of free space (eo = 8.85 x 10 12 C2/[N m2] ), h is Plancks constant (h = 6.626 x 1034 J s), and n is the principal quantum number. (Whew!) The energy of the electron in an isolated hydrogen atom therefore depends only on the value of the quantum number nall the other quantities in the equation are known constants! We can thus simplify this equation (significantly!) by combining a number of the constants in Eq. (9.4) above into a single constant. In particular, it is convenient to define a constant called the Rydberg constant, R, as follows:
![]()
With this definition of the Rydberg constant, it follows that Eq. (9.4) above for the energy of the electron in the nth quantum state can be written in the simpler form
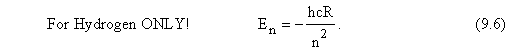 |
Substituting in for the values of the constants h, c, and R in the equation above finally allows us to write the equation for the hydrogen electron energies in the simplest form
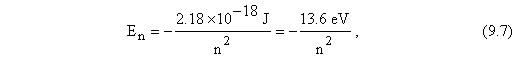 |
where we have shown the energy value in both joules, J, and electron volts, eV ( a small unit of energy often used in atomic and nuclear physics: 1 eV = 1.6 × 1019 J). Note that the energies given by the equation above are all negative this is a result of the fact that the electron is bound to the proton in the atom. Lower energies correspond to states with larger negative numbers for energy values be careful!
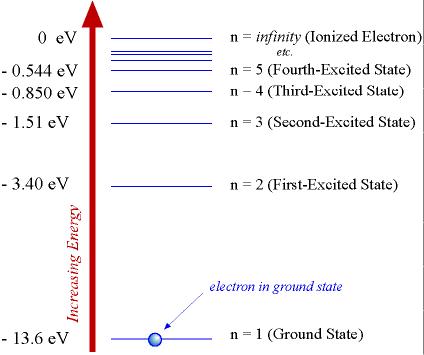
To help us understand more clearly what these energies are telling us, lets compute a few of these energies and the construct an energy-level diagram (see Lecture 8).
The lowest energy given be Eq. (9.7) is the energy for the quantum state having n = 1. Using this value of n in Eq. (9.7) gives us that E1 = 13.6 eV. Since this energy is the lowest energy possible for the electron bound in the hydrogen atom, it must be the ground state energy for the electron, so that the n = 1 quantum state must be the ground state.
Recall from Lecture 8 that the next lowest energy (that is, the first allowed energy above the ground state) is called the first excited-state energy . Inspection of Eq. (9.7) shows that this must correspond to the state with principal quantum number n = 2, for which E2 = 13.6 eV/22 = 3.40 eV. Likewise, the second excited state (n = 3) has an energy given by E3 = 13.6eV/32 = 1.51 eV. (Again, note that these negative numbers are getting closer and closer to zero, so they must be getting larger and largerthey are increasing in energy!) We can therefore construct an energy-level diagram for the electron in the hydrogen atom. Such a diagram is shown in Fig. 9.1 below.
|
We will use this energy-level diagram in the next section to help us understand the emission and absorption spectrum for atomic hydrogen.